Before the Run
How do I update the instrument’s software?
The latest software and instructions for installation can be downloaded from the Pippin Prep support page or the Blue Pippin support page. Please ensure the software is up to date prior to starting a run, as newer versions optimize size selection protocols and improve the user interface.
What is the range/capacity of my cassette?
The size range of our cassettes can be found in our cassette kit reference charts for the Pippin Prep or the Blue Pippin. Most cassettes can accept up to 5 µg DNA per lane, but 1.5% cassettes can only be loaded with up to 2 µg DNA per lane.
I ran out of marker. Can I use marker from a different instrument?
No, markers are unique to each instrument type.
Can I use different lanes in the same cassette for multiple runs?
Yes, as long as current is completely turned off for unused lanes. Cassettes with unused lanes can be resealed (with PCR tape) and reused at a later time up to the expiration date of the cassette. Once current has been applied to a lane, it cannot be used again so make sure to note which lanes have been used.
My instrument does not calibrate correctly.
Ensure that the calibration fixture is placed with the dark side down and that all five LEDs are covered by it. Make sure the lid is completely closed during calibration. If that does not work, try powering down the instrument, unplugging it from the power outlet, and rebooting before repeating the calibration.
My instrument does not pass the conductivity test.
If all lanes fail: Make sure the adhesive plastic covering the cassette is completely removed and that both the cassette and instrument are at room temperature.
If individual lanes fail: Compare buffer levels in the chambers and fill any that are low. Replace buffer in the elution modules to remove any potential air bubbles.
What is the difference between the 4-10 Kb vs2 and 6-10 Kb vs3 high pass protocols?
This document summarizes and illustrates the differences between these high pass protocols.
During the Run
The Photo Signal stops after the internal marker peaks are detected.
This is normal and the machine is behaving as expected. LEDs are only turned on until the markers are detected. They shut off afterwards because they are no longer needed for the run.
When using external marker, I only see signal in the reference lane.
This is normal and the machine is behaving as expected. When an External Marker is used, the instrument only turns on the LED for the reference lane; the LED will remain on in that lane until expected peaks are called and then it too will shut off.
When using Dye Free cassettes, I don’t see a sample trace.
This is normal and the machine is behaving as expected. When using dye free cassettes, no intercalating dye is present so visualization of samples is not expected. Markers are fluorescently tagged so they will be visible.
The instrument has stopped, but the progress bar does not read 100%.
This is normal and the run has completed successfully. The Progress bar only indicates the percent completion of the maximum run time which is determined by the value in the “Run Time” field of the protocol editor. However, the run itself will end at an earlier time if the option “end run when elution is completed” is checked. This box is checked by default, and the value for “Run Time” is longer than it takes for a typical sample to elute, so the progress bar usually does not get to 100% before the run is finished.
After the Run
Does my size selected DNA need to be further purified for next generation sequencing?
DNA eluted from the Dye-Free (DF) cassettes can be immediately used for most downstream NGS applications. However, DNA eluted from ethidium bromide cassettes do need further purification.
My DNA size is lower/higher than expected after a run.
There are three common reasons why this might occur:
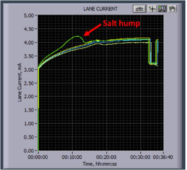
1) Excess salt in the sample (>80mM ionic strength) as indicated by a hump in the lane current (see image below). Salt creates a cloud around the DNA and masks the charge. This causes the DNA to migrate unpredictably during electrophoresis. Further DNA purification should prevent this issue.
Figure 1. A typical hump in lane current due to excess salt in the DNA sample (click to enlarge)
2) The DNA sample interferes with the internal marker. This may happen if the DNA sample has a high concentration of LMW DNA (< 150 bp). The internal marker can be used as an external standard to alleviate this problem.
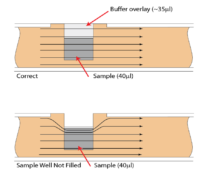
3) The sample well was not completely filled. This can affect the current distribution through the gel (see image below). Ensure that the well is loaded with 40 µL sample and that the buffer overlay reaches the top of the well.
Figure 2. An illustration of the electrophoretic effect on a DNA sample when a sample well is not completely filled (click to enlarge)
My yield is unusually low.
Users should expect an intrinsic yield of 50-80% of the desired size area. If yield is lower, the most common recommendations for improving it are listed in order of effectiveness:
For all runs:
1) Try broadening the size selection.
2) If using tight mode, switch to range mode.
For runs targeting high molecular weight DNA (>2kb, high-pass, etc.):
1) Wait at least 45 min after termination of the run before removing eluted DNA. This allows DNA to detach from the membrane of the elution well.
2) Additional 10-30% yield can often be achieved by rinsing the elution well with 0.1% Tween 20, after removing the initial eluted volume.
3) When removing elution well contents, gently mix the solution by pipetting several times.
None of these answers helped!
If none of these situations apply or if you need assistance with sample recovery, please call customer service at 978.922.1832 or email support@sagescience.com. Including the log files of the affected run in an email will expedite resolution (see below). We also offer free one hour webinars for new users.
Instructions to Export Pippin Log Files
The system automatically saves 4 files per run. Three of them are .png files; one LED calibration, one Continuity Test, and one screen shot that has the name of the protocol and timestamp of the run. The fourth file is a .txt file that has the protocol name/timestamp, you can access these through the File Manager tab.
• Insert a flash drive in to the front USB port of the instrument.
• From the File Manager tab, click on the relevant month/year folder on the left of the screen
• Highlight the relevant files
• Click copy to flash
• Click ‘unmount flash’ before removing the flash drive
• Attach these files to an email to support@sagescience.com